Having worked closely with Chinese authors and pharma clients on manuscript writing project managment, we elaborate on six key areas for Western MedComms agencies to focus on. Check it out now.
As Western life sciences companies explore new markets, China offers significant potential for growth and development. In this article, we delve into four key areas that offer exciting opportunities: aged care, health supplements, early-phase clinical trials, and medical manuscript writing support. Check it out now.
This article covers effective tips to edit and revise a medical translation to deliver to a client, as well as from a client side. Read on to find out!
In this post about Chinese business culture, we cover: the curious case of company seal; How to respectfully address your Chinese business partner; and more.
This article provides a glimpse of important Chinse festivals and Chinese culture. If you get a tiny bit out of it, and practice it with your Chinese clients, they will for sure appreciate your efforts!
A crucial regulation on China health food is updated. Here AMC compiles its important sections in English, especially where overseas health food companies are concerned. We also provide expert interpretation and future directions. Check it out!
Here are the current China advertising guidelines for drugs, medical devices, health food and food for special medical purposes. Check it out!
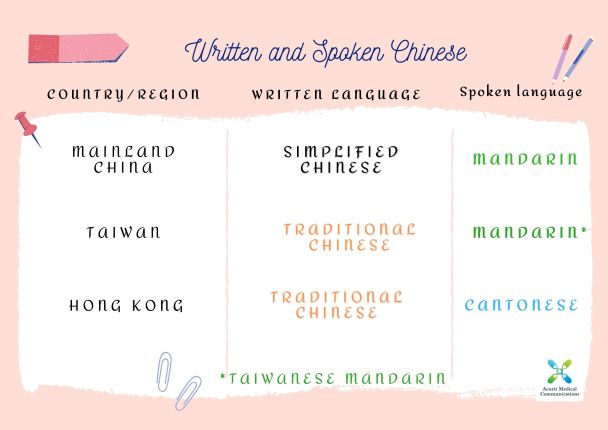
Simplified Chinese and Traditional Chinese are the two written Chinese characters. Mandarin is the official spoken language in mainland China. Click to red more.
I have been working from home for about five years. With what is going on in the world, many things become our new normal, including working from home. Read the tips here.
The China “Provisions for Drug Registration” have been issued and will be implemented on July 1, 2020. The new Provisions are the first revision in 13 years. Here we highlight and analyse changes from the previous version.